New chemical tools and technologies to study cellular redox biology
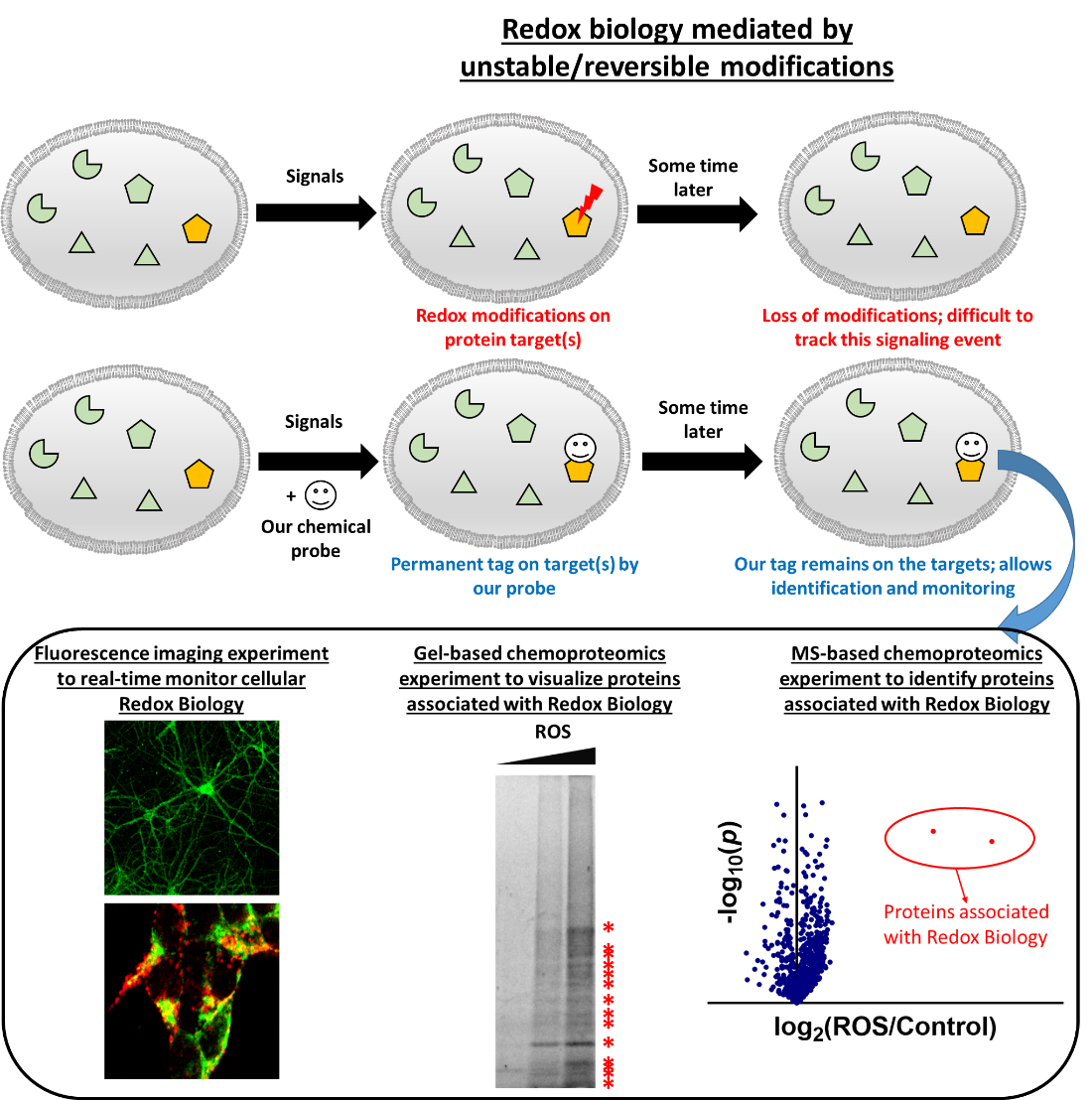
Cellular redox biology is governed by interesting classes of redox-active molecules, which can mediate reduction/oxidation of biomolecules. Notably examples of redox-active molecules include reactive oxygen species (ROS) such as hydrogen peroxide (H2O2) and superoxide (O2–), and hydrogen sulfide (H2S) which belongs to reactive sulfur species (RSS) family. These redox-active molecules were once classified as detrimental compounds in biology and known to contribute to serious disease development such as cancer and neurodegenerative disorders. Yet, recently these redox-active molecules are found to be essential to life and can serve as important signaling molecules. This is mainly through their Redox Biology, which induces reduction/oxidation of biomolecules and subsequently changes their functions, activities and downstream signals.
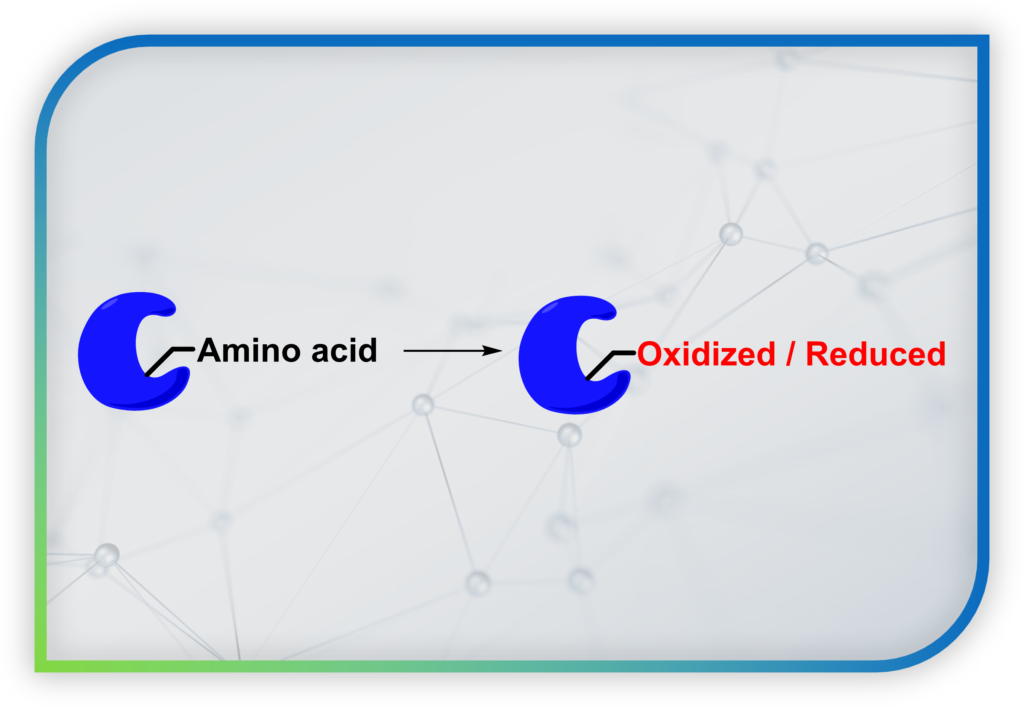
Despites nowadays there are better ideas on redox signaling and its biological functions, the molecular mechanism on initiation, transduction and regulation of redox signaling, as well as the identity of proteins involved in the signaling process, remain insufficiently understood. This is because redox modifications of proteins are reversible, dynamic and unstable. In contrast to protein phosphorylation/dephosphorylation which are more stable modifications, there are no good conventional biochemical and biological experiments to study protein redox modifications and hence Cellular Redox Biology.
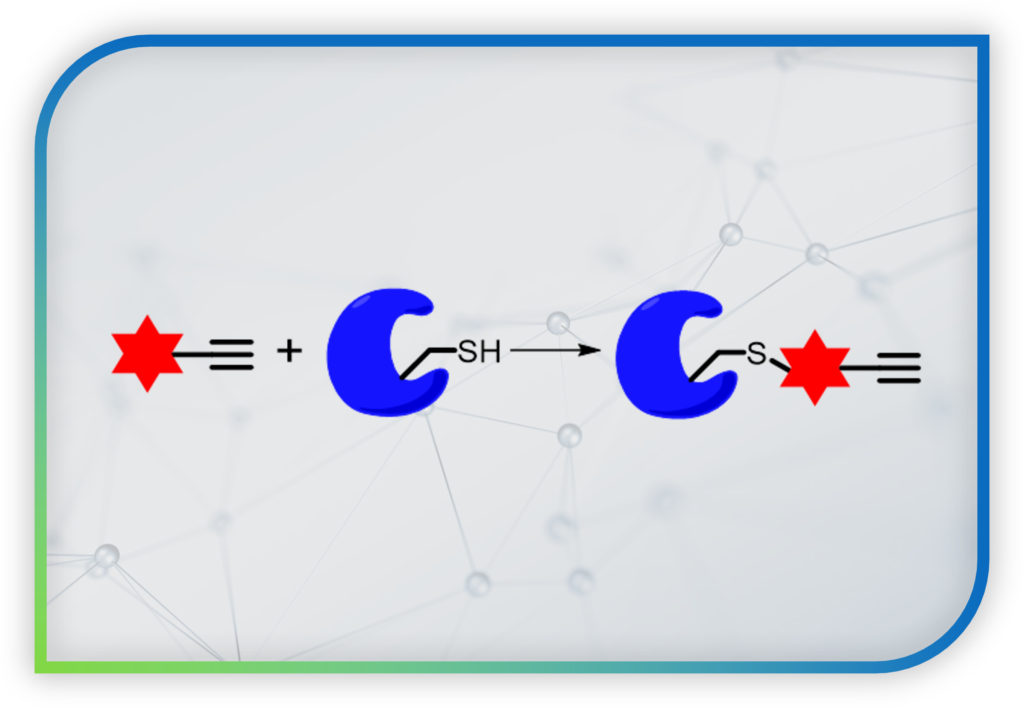
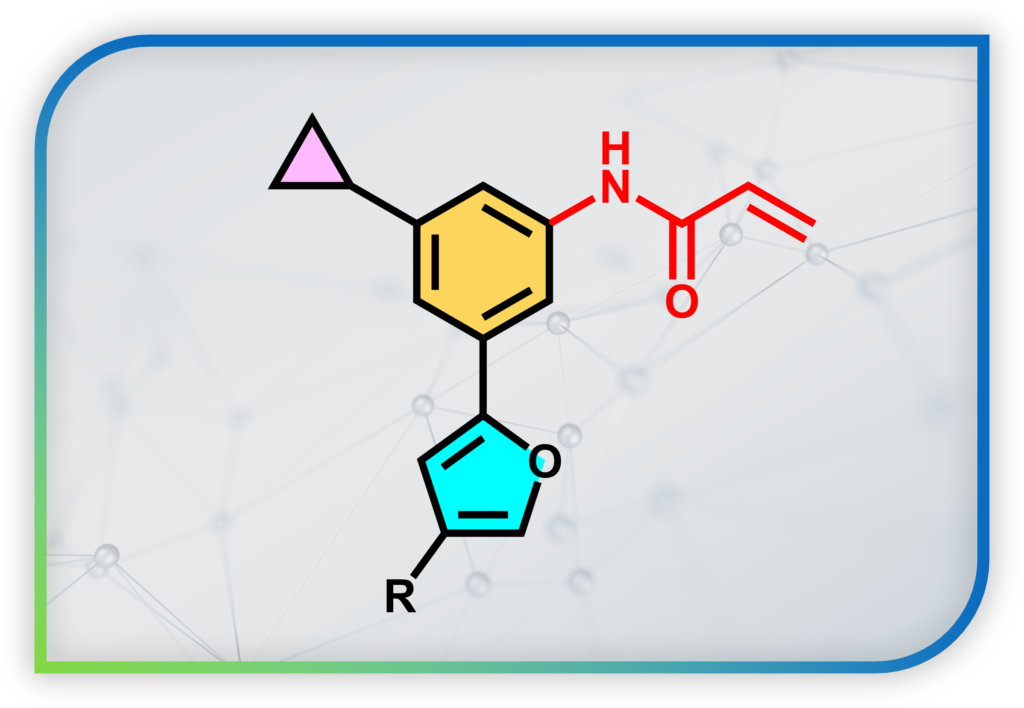
To overcome this challenge, our lab is developing new chemical probes that can: (1) trap “reversible redox modification” on proteins associated with Redox Biology into “permanent covalent tag”. This allows us to identify the proteins by proteomics and mass spectrometry (MS)-based experiments. (2) We can also real-time visualize redox signaling events by turning the permanent covalent tag into a fluorescent one. This enables fluorescence imaging with superior spatial resolution.